Abstract
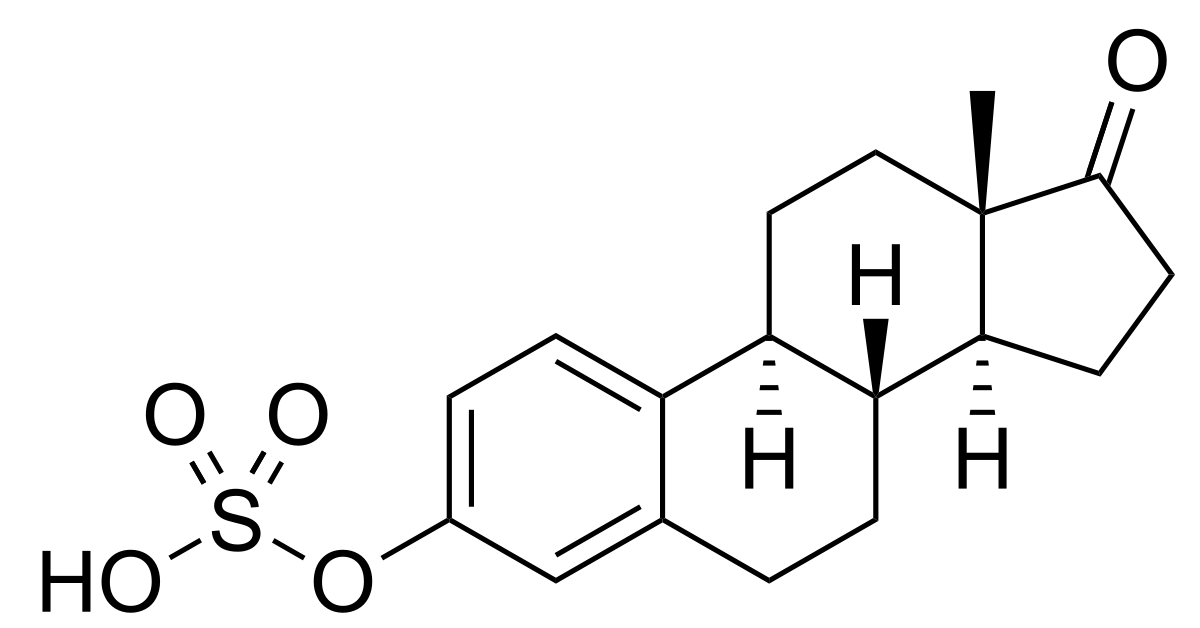
Conjugated equine estrogen therapy is the most common hormone replacement strategy used to treat postmenopausal women. However, the ability of an individual conjugated equine estrogen to modulate NO production and, therefore, to induce cardiovascular protection is largely unknown. The effects of equine and naturally occurring estrogens on NO generation were evaluated in human aortic endothelial cells by measuring in vivo NO production, as well as NO synthase (eNOS) activity and expression. The transcriptional activity on the eNOS gene was determined by the ability of estrogen receptors (alpha and beta) to activate the eNOS promoter and induce transcription. Docking and molecular dynamics simulations were used to study structural features of the interaction between estrogenic compounds and estrogen receptor-alpha. After 24 hours of incubation, we found that estrone upregulated NO production almost as effectively as estradiol by increasing eNOS activity and expression. However, the effect of equine estrogens (equilin, equilenin, and their metabolites) were marked decreased. eNOS promoter activity by equine estrogens was 30% to 50% lower than the naturally occurring estrogens. Computational analysis of estrogen molecules revealed that position 17 and the saturation of estrogenic compounds in ring B are important determinants for estrogen receptor-alpha transcriptional activity. Equine estrogens increase NO production less effectively than naturally occurring estrogens, partially because of their lesser ability to activate the eNOS promoter and induce transcription. Differences in NO production by different estrogens may account for the differences in cardiovascular benefits achieved by the distinct estrogen replacement therapies.
Novensa L, Selent J, Pastor M, Sandberg K, Heras M, Dantas AP.